Measuring Hydrogen
The focus is on development of spectroscopic techniques that allow one to gain insight into the interplay between electronic, optical, magnetic and chemical properties of matter and hydrides. The current areas of expertise include, but are not limited to, table-top ambient pressure XPS for studying metallic and organic membranes exposed to hydrogen and other gases, and magneto-optical Faraday and Kerr effect set-up for analysis of photo- and electrochemically induced electronic structures of water splitting catalysts.
Chemical analysis of CO2 reduction
A range of special analysis tools are utilized to characterize processes generating fuel from CO2 and hydrogen. We make use of our state-of-the-art facilities establishing collaborations and proving consulting for academia and industry. The element in focus is hydrogen as its analysis faces specific challenges. Here, we collaborate with a number of large research facilities (PSI-SINQ, RAL-ISIS, ORNL-SNS) providing neutrons as a selective probe for hydrogen in matter.
Recent highlights are the development and utilization of neutron imaging to observe the CO2 reduction in real-time in collaboration with the Laboratory for Neutron Scattering and Imaging at PSI. The movie below shows the diffusion of water into mm-sized zeolite beads. Further fascinating results are found in the recent publication in Journal Physical Chemistry.
The video shows a H2O absorption experiments into mm-sized zeolite beads, visualized using neutron imaging. Image size is 4x5 mm2, time step is 1 frame per minute (fpm).
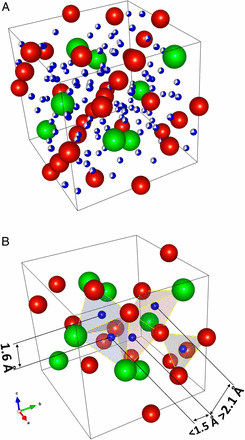
Inelastic neutron scattering evidence for anomalous H–H distances in metal hydrides
Hydrogen in metals alters the electronic structure of such materials and hence modifies the physical and chemical properties. In conventional transition metal hydrides containing atomic hydrogen, the minimum hydrogen–hydrogen distances are around 2.1 Å under ambient conditions (Switendick criterion). Although hints of H–H distances below 2.1 Å in AB2 alloys have been reported, evidence is inconclusive as hydrogen positions are difficult to locate by diffraction techniques. Here, inelastic neutron scattering is used as a local probe of the hydrogen interactions together with electronic structure modeling of a well-studied and prototypical metal hydride ZrV2Hx. The results provide evidence for anomalous hydrogen–hydrogen distances as short as 1.6 Å. The findings provide insights leading to the creation of materials with properties such as very high Tc superconductivity and other quantum behaviors.
Ultra-High Pressure Hydride with XPS
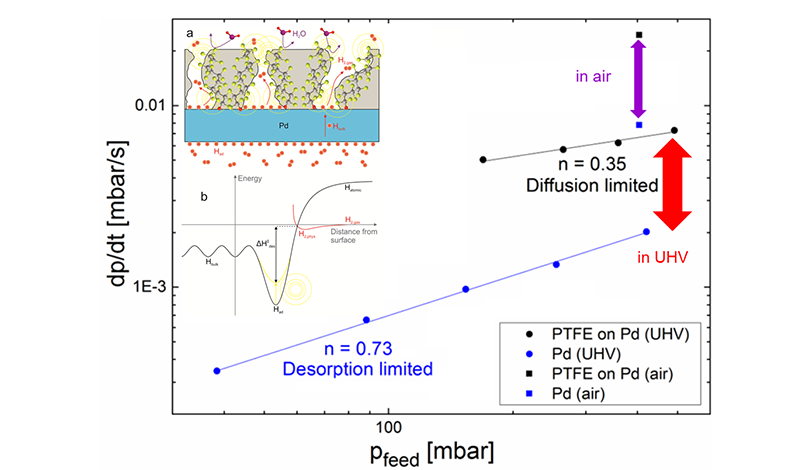
The measurement and control of hydrogen at surface is challenging. In this project, funded by SNSF (grant number 200021_172662) we develop a novel measurement method being able to unravel the mechanisms driving the hydrogen – matter interaction taking place in hydrogen storage, hydrogen selective membranes, and the catalysis of hydrogenation reactions. Although apparently simple, a direct photoemission measurement of the hydrogen induced changes of the electronic structure, which are the origin of the binding of hydrogen with metals, is possible in a few cases only. The reason for this is purely technical: the electronic structure of hydrogen chemisorbed to surfaces can be measured using standard surface science techniques, because the required hydrogen pressure is compatible with the UHV technology. However, processes relevant for energy conversion and storage take place at several atmospheres hydrogen pressure; and thus valuable information on these systems is not accessible by commonly used surface science methods due to their incompatibility with high pressures.
The present project relies on a membrane approach for high pressure XPS under development in our laboratory. The method is based on a new type of specimen holder, which is a metallic, hydrogen permeable membrane fed on one side with a high hydrogen pressure and exposed on the other side to the X-ray beam at UHV-pressures. In first papers, we introduced the fundamental idea and demonstrated the feasibility of the method in some well-studied cases, paving the way for its use on relevant questions in energy storage. In this project, we want to utilize the membrane approach to prepare and measure in-situ various ionic and intermetallic hydrides as a function of the chemical potential of hydrogen, which will deliver insightful knowledge on the electronic structure of hydrides. In a second task, we want to elucidate the mechanism of the enhanced hydrogen desorption from hydrogen membranes upon application of polymer coatings. Finding the physical origin of the remarkable effect may help to functionalize also hydrogen permeable metals other than Pd.
Finally, the catalytic formation/decomposition of the C-H bond is one of the main challenges in synthetic chemistry, due to the strong covalent nature of this bond. The (de-) hydrogenation reactions of hydrocarbons, which take place at several tens of bar, are catalysed by Pd, Ru, or similarly expensive and scarce elements. Experiments on the hydrogenation of unsaturated hydrocarbons using the Pd-membrane mimic the technical process, so that information on the reaction can be drawn. Finally, we want to explore the possibility of combining different spectroscopy methods (X-ray absorption spectroscopy and Raman spectroscopy) with the membrane approach.
LightCheC project in collaboration with the University of Zurich
“Laser membrane photoemission and magneto-optical spectroscopy shining light on solar water splitting” (see also https://www.empa.ch/web/s502/catalytic-methanation) We are heading for a comprehensive understanding of the electronic structure and its dynamics of materials for solar water splitting. The work may be divided into two parts: the investigation of homo-geneous and heterogeneous solar water splitting by time resolved magneto-optical spectroscopy (trMOKE) and by laser-induces photoemission. For the latter, we are developing a pulsed laser-driven plasma UV source and membrane-based tabletop photoemission set-up. Thus, the method provides information on in-situ probing of the electronic changes in the catalyst over the course of the reaction and offers an alternative to the expensive and often inaccessible synchrotron sources. The magnetic properties of materials are very sensitive to the electronic properties and thus serve as a probe for the changes induced by light and adjacent chemical processes in photocatalytic reactions. Thus, we take advantage of the trMOKE approach for studying magnetic and optical properties of homogeneous and heterogeneous systems in transmission and reflection geometries. Apart from empirical investigations, we model the obtained data using the established models for anticipating the activity of catalytic sur-faces (e.g., the Norskov model). We are aiming at extending these models by the time dimension as needed in photo-catalysis.