Lithium-ion and sodium-ion batteries
Lithium-ion batteries with liquid electrolytes remain the dominant technology for portable electronics, electric vehicles, and stationary energy storage systems, owing to their high energy density, excellent cycling stability, and low cost enabled by gigafactory-scale production. Recent industry trends include the differentiation of cell chemistries tailored to specific applications, the incorporation of silicon into anodes to further boost energy density and driving range, and improvements in power performance to support increasingly faster charging. From a regulatory perspective, growing emphasis is placed on sustainability, near-sourcing of raw materials and cells, and enhanced safety. Sodium-ion batteries are emerging as a promising alternative, particularly for stationary energy storage but also low-cost mobility applications, offering advantages such as abundant raw materials and improved safety.
Our research focuses on electrolytes and interphases that stabilize lithium-ion batteries with high-capacity nickel-rich layered LiNixMnyCo(1-x-y)O2 (x>0.8) or high-voltage spinel LiNi0.5Mn1.5O2 cathodes [1-3]. By combining suitable solvents, lithium salts, and additives, advanced electrolytes can signifi-cantly improve the cycling stability, reduce flammability, or increase temperature range of use (a).
We also have strong expertise in aqueous electrolytes. Using high salt concentrations, the electrochemical stability window can be expanded, enabling aqueous batteries with higher cell voltages (b, c).
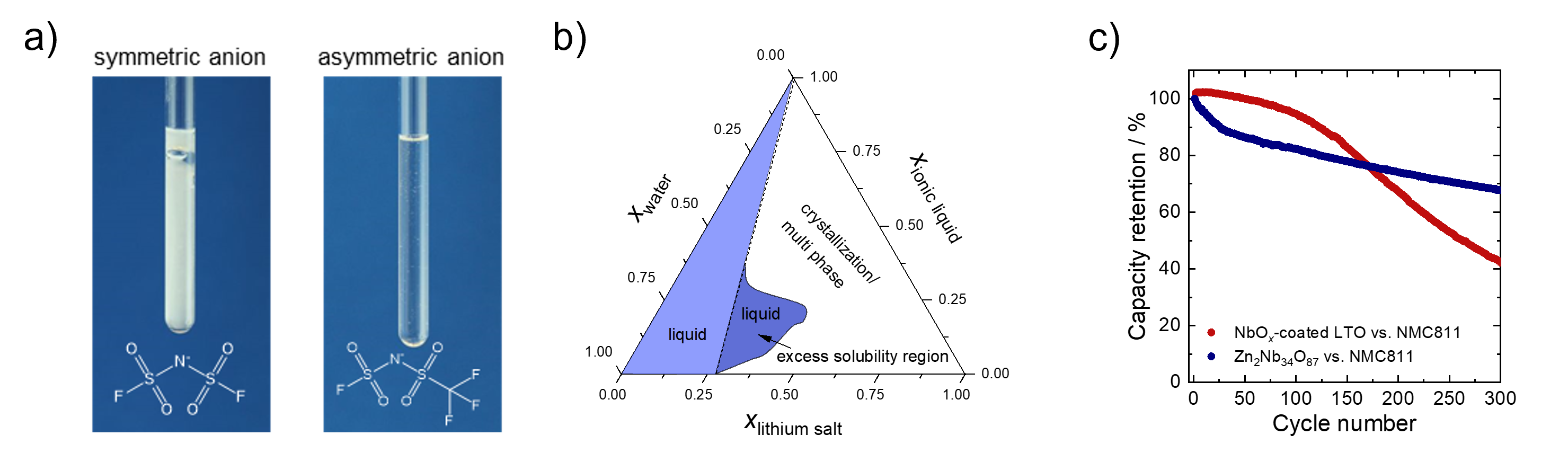
(a) Asymmetric anions can enable highly concentrated electrolytes that remain liquid in a wide tem-perature range by suppressing crystallization. (b) Phase diagram for a water-ionic liquid-lithium salt electrolyte that enables very high lithium-salt concentration. (c) Cycling performance of NMC811-based full cells containing NbOx-coated Li4Ti5O12 (LTO, in red) or Zn2Nb34O87 (in blue) as anode mate-rial. The cell contains a water-in-salt/ionic liquid hybrid electrolyte.
Selected publications
[1] Y. Kim, A. Moysiadou, K. Egorov, D. Rentsch, C. Battaglia, A strategy to prevent fluorine-induced transition metal dissolution in lithium-ion batteries, Advanced Energy & Sustainability Research 2025, 2500194, https://doi.org/10.1002/aesr.202500194
[2] S. Liu, M. Becker, Y. Huang-Joos, H. Lai, G. Homann, R. Grissa, K. Egorov, F. Fu, C. Battaglia, R.-S. Kühnel, Multifunctional additive ethoxy(pentafluoro)cyclotriphosphazene enables safe carbonate electrolyte for SiOx-graphite/NMC811 batteries, Batteries & Supercaps 2023, 6, e202300220, https://doi.org/10.1002/batt.202300220
[3] K. Egorov, W. Zhao, K. Knemeyer, A. N. Filippin, A. Giraldo, C. Battaglia, Mitigating first-cycle ca-pacity losses in NMC811 via lithicone layers grown by molecular layer deposition, ACS Applied Materials Interfaces 2023, 15, 20075, https://doi.org/10.1021/acsami.2c23158
[4] M. Becker, F. Bernasconi, K. Egorov, E. Svaluto-Ferro, R.-S. Kühnel, C. Battaglia, Niobium oxide an-ode materials with suppressed activity toward hydrogen evolution reaction for aqueous batteries, Energy Storage Materials, 2024, 71, 103613, https://doi.org/10.1016/j.ensm.2024.103613
[5] M. Becker, W. Zhao, F. Pagani, C. Schreiner, R. Figi, W. Dachraoui, R. Grissa, R.-S. Kühnel, C. Battaglia, Understanding the stability of NMC811 in lithium-ion batteries with water-in-salt elec-trolytes, ACS Applied Energy Materials, 2022, 5, 11133, https://doi.org/10.1021/acsaem.2c01722
[6] M. Becker, D. Rentsch, D. Reber, A. Aribia, C. Battaglia, R.-S. Kühnel, The hydrotropic effect of ionic liquids in water-in-salt electrolytes, Angewandte Chemie International Edition, 2021, 60, 14100, https://doi.org/10.1002/anie.202103375
[7] D. Reber, O. Borodin, M. Becker, D. Rentsch, J. H. Thienenkamp, R. Grissa, W. Zhao, A. Aribia, G. Brunklaus, C. Battaglia, R.-S. Kühnel, Water/ionic liquid/succinonitrile hybrid electrolytes for aque-ous batteries, Advanced Functional Materials, 2022, 32, 2112138, https://doi.org/10.1002/adfm.202112138
[8] D. Reber, R. Grissa, M. Becker, R.-S. Kühnel, C. Battaglia, Anion selection criteria for water-in-salt electrolytes, Advanced Energy Materials, 2021, 2002913, https://doi.org/10.1002/aenm.202002913
[9] R.-S. Kühnel, D. Reber, C. Battaglia, A high-voltage aqueous electrolyte for sodium-ion batteries
ACS Energy Letters, 2017, 2, 2005, https://doi.org/10.1021/acsenergylett.7b00623
Funding
Swiss Federal Office of Energy, Innosuisse, Swiss State Secretariat for Education, Research, and Inno-vation, Horizon 2020, Horizon Europe, Göhner Foundation, Metrohm Foundation, industry projects.


Next-generation lithium-ion batteries, kick-off for European battery research project SeNSE, https://www.empa.ch/web/s604/sense-kickoff-meeting
Successful European battery project, from raw material to an (almost) finished car battery, https://www.empa.ch/web/s604/sense-better-batteries-for-electric-cars
Inexpensive and stable, the salt water battery, https://www.empa.ch/web/s604/salt-water-battery
-
Share
