On the road to artificial photosynthesis
Nature knows how to do it – as does research, in principle
The need for a sustainable energy supply is now a hot topic. Companies are investing in it, and scientists around the globe are looking for alternatives to the production of energy from oil and gas. One such project, which has been running at the University of Zurich since the beginning of 2013, is the large-scale research project, LightChEC. As the name suggests, this university research project focuses on "turning solar energy into chemical energy". The researchers involved are aiming to refine artificial photosynthesis to make it suitable as a means for supplying energy to industry and society. Three Empa scientists, Andreas Borgschulte, Karl-Heinz Ernst and Andreas Züttel, are carrying out research as part of LightChEC. Ernst and Züttel are also on the Steering Committee.
Nature provides the blueprint
Trees, shrubs, grasses and algae have been using photosynthesis since time immemorial. To do this, they create the natural pigment chlorophyll in their cells. The parts of the plant containing chlorophyll convert carbon dioxide (CO2) and water into oxygen and glucose with the aid of sunlight. The glucose acts as the plant's energy source and the oxygen is released. This is why forests are often referred to as "green lungs".
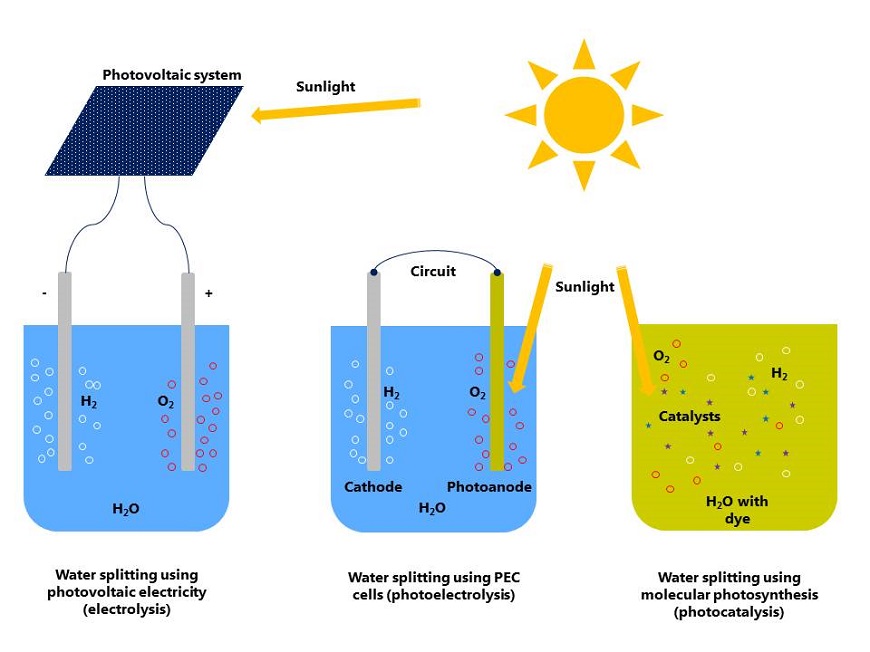
Artificial photosynthesis aims to mimic its natural role model. In essence, this is less about the production of glucose, but rather "simply" the separation of water into hydrogen and oxygen using sunlight. The hydrogen obtained can then be used either directly as a fuel, for example, to power fuel cell vehicles, or it can be used to synthesise hydrocarbons, such as methane – the main component of natural gas –, ethane, propane or even (liquid) octane.
The researchers are currently pursuing various ideas for copying natural photosynthesis, as Empa researcher Andreas Borgschulte is doing. In the simplest approach, water is split into hydrogen and oxygen by electrolysis using solar-generated electricity. The harvesting of sunlight using solar cells and the electrolysis of water are processes that (can) in principle be carried out separately. However, at the microscopic level, they can be combined, as Andreas Borschulte explains. This is referred to as photoelectrolysis in what are known as photoelectrochemical cells, abbreviated to PEC cells. Researchers from Empa unveiled a PEC cell back in 2014. This can be envisaged as a water-filled vessel with a photoanode and a counter electrode. The photoanode absorbs sunlight and provides the electricity to split the water. Therefore, the process does not involve solar cells. PEC cells currently have an efficiency of around five percent. This means that one-twentieth of the solar energy "captured" is converted into chemical energy – hydrogen.
Method with potential for the future
Scientists working on the LightChEC project recently succeeded in imitating the whole process in a molecular system in an aqueous solution, thereby removing the need for the electrodes used in PEC cells. With this method, known as photocatalysis, researchers have come a significant step closer to the natural model compared to PEC cells.
However, the efficiency of this method, which was developed as part of LightChEC, is not yet able to match the efficiency of PEC cells. Therefore, the task now is to optimise the various (individual) steps, for example, by further improving the chlorophyll-like dyes or the water-splitting catalysts.
For the time being, however, in addition to their relatively high efficiency, PEC cells offer another advantage: they can already be used today. Also the electrolysis with photovoltaic electricity is already applied, for instance in Empa's "move" mobility demonstrator: with this, Empa aims to show how excess electricity from photovoltaic and hydroelectric power plants can be used efficiently for sustainable mobility, such as for directly charging electric cars or for producing and storing hydrogen or methane.
The search for better catalysts
Empa researchers have already developed the corresponding method to a large extent, but wish to improve it further. This includes looking for ever better catalysts. These determine, for example, how efficiently CO2 and hydrogen react to form methane. Using the latest analysis techniques, the Empa researchers want to better understand the mechanisms of these catalysts, i.e. the molecular reaction mechanism, thereby helping to make the production of energy via artificial photosynthesis commercially viable at some point in the future.
Further information
Andreas Borgschulte
Advanced Analytical Technologies
Phone +41 58 765 4639
Editor / Media contact
Reto Zanettin
Communications
redaktion@empa.ch
Water splitting using PEC cells requires efficient and stable photoactive materials. Research is focusing in particular on materials for the photoanode, which harvests sunlight. However, all the materials used up to now have entailed efficiency losses caused by various mechanisms, thus reducing the efficiency of the photoelectrolysis.
Recently, a research group from Empa's Laboratory for Materials for Energy Conversion presented a strategy for eradicating these losses: the researchers, led by Simone Pokrant, coated the photoanode multiple times with different materials, so that each layer eliminated a loss mechanism. The combination of all the layers significantly increased the overall efficiency of the PEC cell. The process used by the Empa team is also relatively easy to scale up, thus bringing its large-scale application a step closer.
At the start of May 2016, project leader Simone Pokrant was appointed as a professor at Saarland University of Applied Sciences. In this role, she will carry out research into electrochemical energy storage systems from June, including continuing with her work on PEC cells. At Empa, Simone Pokrant worked since 2012 on developing materials for photoelectrochemical water splitting, batteries and luminescent dyes.
Literature
S Landsmann, Y Surace, M Trottmann, S Dilger, A Weidenkaff, S Pokrant (2016) Controlled Design of Functional Nano-Coatings: Reduction of Loss Mechanisms in Photoelectrochemical Water Splitting, Applied Materials & Interfaces, DOI: 10.1021/acsami.6b01129
-
Share
